Summary
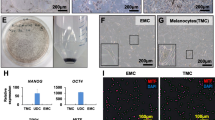
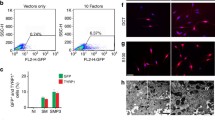
Vitiligo is an enigmatic pigmentary disorder of the skin. Factors potentially involved in the progressive loss of melanocytes from the basal layer of the epidermis include genetically determined aberrancies of the vitiligo melanocyte. It follows that analysis of melanocytes cultured from vitiligo donors can contribute to a further understanding of the etiopathomechanism. A setback for vitiligo research has been the limited availability of vitiligo-derived melanocytes. To overcome this limitation, we have generated a vitiligo melanocyte cell line according to a protocol established previously for the immortalization of normal human melanocytes. Vitiligo melanocytes Ma9308P4 were transfected with HPV16 E6 and E7 genes using the retroviral construct LXSN16E6E7. Successful transformants were selected using geneticin and subsequently cloned to ensure genetic homogeneity. The resulting cell line PIG3V has undergone more than 100 cell population doublings ince its establishment as a confluent primary culture, whereas untransfected melanocytes derived from adult skin senesce after a maximum of 50 population doublings. Cells immortalized by this transfection procedure retain lineage-specific characteristics and proliferate significantly faster than parental cells. In this study, the phenotype of PIG3V resembled melanocytes rather than melanoma cells in culture. Tyrosinase was processed properly and melanosomes remained pigmented. Importantly, ultrastructural characterization of PIG3V cells revealed dilated endoplasmic reticulum profiles characteristic of vitiligo melanocytes. An explanation for this dilation may be found in the retention of proteins with molecular weight of 37.5, 47.5, and 56.5 kDa, as determined by gel electrophoresis of microsomal proteins isolated from radiolabeled cells.
Similar content being viewed by others
References
Boissy, R. E.; Liu, Y. Y.; Medrano, E. E.; Nordlund, J. J. Structural aberration of the rough endoplasmic reticulum and melanosome compartmentalization in long-term cultures from vitiligo patients. J. Investig. Dermatol. 97:395–404; 1991.
Boissy, R. E.; Zhao, Y.; Gahl, W. A. Altered protein localization in melanocytes from Hermansky-Pudlak syndrome: support for the role of the HPS gene product in intracellular trafficking. Lab. Invest. 78:1037–1048; 1998.
Galardi, E.; Mehrigan, A. H.; Hashimoto, K. Ultrastructural study of vitiligo. Int. J. Dermatol. 32:269–271; 1993.
Halaban, R.; Alfano, F. D. Selective elimination of fibroblasts from cultures of normal human melanocytes. In Vitro 20:447–450; 1984.
Halbert, C. L.; Demers, G. W.; Galloway, D. A. The E7 gene of human papillomavirus type 16 is sufficient for immortalization of primary human epithelial cells. J. Virol. 65:473–478; 1991.
Hawley-Nelson, P.; Vousden, K. H.; Hubbert, N. L., et al. HPV16 E6 and E7 proteins cooperate to immortalize human foreski keratinocytes. EMBO J. 8:3905–3910; 1989.
Im, S.; Hann, S. K.; Kim, H. I., et al. Biologic characteristics of cultured human vitiligo melanocytes. Int. J. Dermatol. 33:556–562; 1994.
Jimbow, K.; Chen, H.; Park, J. Molecular mechanisms for TRP-1 involvement in vitiligo/leukoderma [abstract]. Pigment Cell Res. S5:234; 1996.
Jones, H. W. Record of the first physician to see Henrietta Lacks at the Johns Hopkins Hospital: history of the beginning of the HeLa cell line. Am. J. Obstet. Gynecol. 176:S227-S228; 1997.
Karnovsky, M. J. A formaldehyde-glutaraldehyde fixative of high osmolaity for use in electron microscopy [abstract]. J. Cell. Biol. 27:137; 1965.
Karnovsky, M. J. Use of ferrocyanide-reduced osmium tetroxide in electron microscopy [abstract]. J. Cell. Biol. 51:146; 1971.
Katano, M.; Saxton, R. E.; Cochran, A. J.; Irie, R. F. Establishment of an ascitic human melanoma cell line that metastasizes to lung and liver in nude mice. J. Cancer Res. Clin. Oncol. 108:197–203; 1984.
Kim, P.S.; Arvan, P. Endocrinopathies in the family of endoplasmic reticulum storage diseases: disorders of protein trafficking and the role of ER molecular chaperones. Endocr. Rev. 19:173–202; 1998.
Kiyono, T.; Foster, S. A.; Koop, J. I., et al. Both Rb/p16INK4A inactivation and telomerase activity are required to immortalize human epithelial cells. Nature 396:84–88; 1998.
Kovasc, S. O. Vitiligo. J. Am. Acad. Dermatol. 38:647–666; 1998.
Le Poole, I. C.; Boissy, R. E. Vitiligo. Semin. Cutan. Med. Surg. 16:3–14; 1997.
Le Poole, I. C.; van den Berg, F. M.; van den Wijngaard, R. M. J. G. J., et al. Generation of a human melanocyte cell line by introduction of HPV16 E6 and E7 genes. In Vitro 33:42–49; 1997.
Le Poole, I. C.; van den Wijngaard, R. M. J. G. J.; Westerhof, W., et al. Presence or absence of melanocytes from vitiligo lesions: an immunohistochemical investigation. J. Investig. Dermatol. 100:816–822; 1993.
Majumder, P.; Nordlund, J. J.; Nath, S. K. Pattern of familial aggregation of vitiligo. Arch. Dermatol. 129:994–998; 1993.
Marquardt, T.; Helenius, A. Mistolding and aggregation of newly synthesized proteins in the endoplasmic reticulum. 117:505–513; 1992.
McKeehan, W. L.; Barnes, D.; Reid, L., et al. Frontiers in mammalian cell culture. In Vitro Cell. Dev. Biol. 26:9–23; 1990.
Melbije, M.; Frisch, M. The role of pappilomaviruses in anogenital cancers. Semin. Cancer Biol. 8:307–313; 1998.
Moellmann, G.; Klein-Angerer, S.; Scollay, D. A., et al. Extracellular granular material and degeneration of keratinocytes in the normally pigmented epidermis of patients with vitiligo. J. Investig. Dermatol. 79:321–330; 1982.
Munger, K.; Scheffner, M.; Huibregtse, J. M.; Howley, P. M. Interactions of HPV16 E6 and E7 oncoproteins with tumour suppressor gene products. Cancer Surv. 12:197–217; 1992.
Nath, S. K.; Majumder, P. P.; Nordlund, J. J. Genetic epidemiology of vitiligo: multilocus recessivity cross-validated. Am. J. Hum. Genet. 55:981–990; 1994.
Nicchitta, C. V.: Migliaccio, C.; Blobel, G. Biochemical assembly of the membrane components that mediate chain targeting and translocation. Cell 65:587–598; 1991.
Nordlund, J. J. The epidemiology and genetics of vitiligo. Clin. Dermatol. 15:875–878; 1997.
Puri, N.; Mojamdar, M.; Ramaiah, A. In vitro growth characteristics of melanocytes obtained from adult normal and vitiligo subjects. J. Investig. Dermatol. 88:434–438; 1987.
Ramaiah, A.; Mojamdar, M.; Amarnath, V. M. Vitiligo in the SSK community of Bangalore. Indian J. Dermatol. Venereol. Leprol. 54:251–254; 1988.
Tomita, Y.; Yamamoto, H.; Sato, C., et al. Efficient culturing of human melanocytes from suction blishters. Tokohu J. Exp. Med. 147:219–320; 1985.
Xu, A.; Bellamy, A. R.; Taylor, J. A. BiP (GRP78) and endoplasmin (GRP94) are induced following rotavirus infection and bind transiently to an endoplasmic reticulum-localized virion compartment. J. Virol. 72:9865–9872; 1998.
Zhang, J. X.; Braakman, I.; Matlack, K. E.; Helenius, A. Quality control in the secretory pathway: the role of calreticulin, calnexin and BiP in the retention of glycoproteins with C-terminal truncations. Mol. Biol. Cell 8:1943–1954; 1997.
Zhao, H.; Boissy, Y. L.; Abdel-Malek, Z.; King, R. A.; Nordlund, J. J.; Boissy, R. E. On the analysis of the pathophysiology of Chediak-Higashi syndrome. Defects expressed by cultured melanocytes. Lab. Invest. 71:25–34; 1994.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Le Poole, I.C., Boissy, R.E., Sarangarajan, R. et al. PIG3V, an immortalized human vitiligo melanocyte cell line, expresses dilated endoplasmic reticulum. In Vitro Cell.Dev.Biol.-Animal 36, 309–319 (2000). https://doi.org/10.1290/1071-2690(2000)036<0309:PAIHVM>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1071-2690(2000)036<0309:PAIHVM>2.0.CO;2